(Author / F.S.JI Reviewed by / S.Z.YANG) Hepatocellular carcinoma (HCC) is characterized by prominent fibrotic deposition and an immunosuppressive microenvironment. Although immune checkpoint blockade (ICB) has become a first-line therapy for advanced HCC, the objective response rate remains below 30%, highlighting an urgent need to overcome therapeutic resistance. Hepatic stellate cells, the liver-specific stromal cells, can differentiate into cancer-associated fibroblasts (CAFs) with marked phenotypic heterogeneity. The excessive collagen deposition and extracellular matrix (ECM) crosslinking mediated by CAFs form a substantial barrier to immune cell infiltration. However, the heterogeneity, spatial organization, and mechanistic roles of CAF subtypes in immunotherapy resistance remain poorly understood.
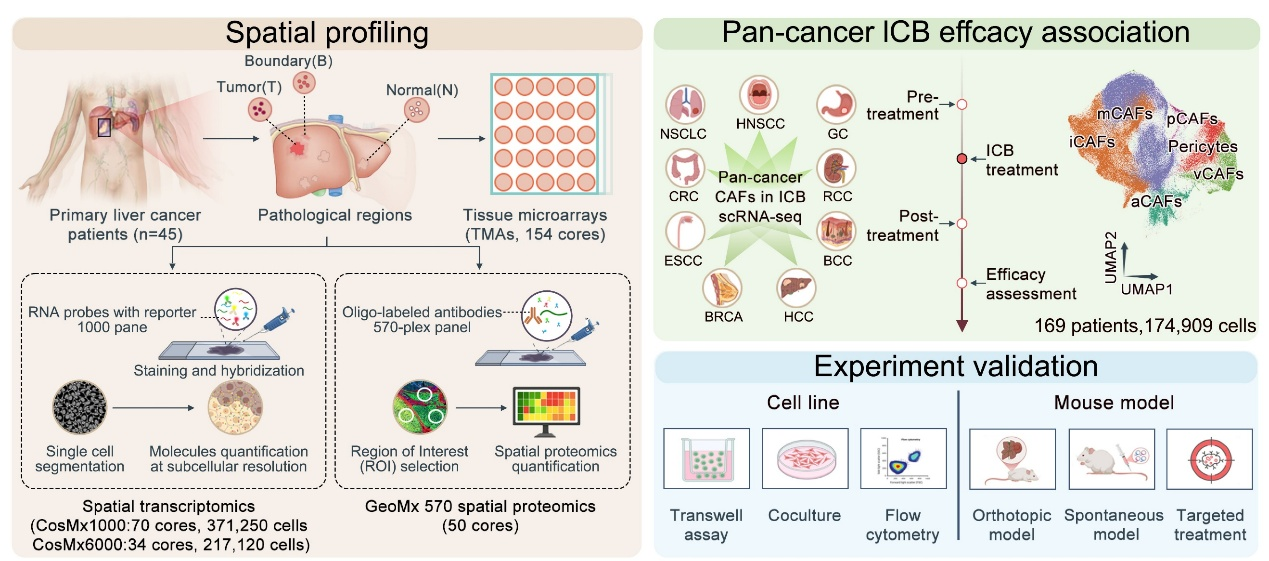
Recently, a team led by Academician Dong Jiahong and Dr. Yang Shizhong at Beijing Tsinghua Changgung Hospital applied CosMx SMI spatial multi-omics technology, combined with the in-house developed pan-cancer single-cell immunotherapy database scICB, to systematically profile the spatial molecular landscape of tumor core, invasive margin, and adjacent regions from 46 primary HCC patients. This study, for the first time at spatial resolution, identifies the NOTCH3 signaling axis as a central regulator linking fibrosis to immunotherapy resistance in HCC.
The study revealed pronounced functional polarization and spatial stratification of CAFs in the HCC microenvironment, with inflammatory CAFs (iCAFs) and myofibroblastic CAFs (mCAFs) as the two dominant subtypes. mCAFs were enriched in the tumor core and invasive margin, and were strongly associated with increased collagen deposition, immune exclusion, and poor prognosis. In contrast, iCAFs were predominantly localized in adjacent non-tumor regions and correlated with more favorable outcomes. Integration with over 300 patients and more than 800 immunotherapy samples from the scICB database further demonstrated that mCAF enrichment is significantly associated with resistance to immunotherapy, whereas higher iCAF proportions correlate with clinical response.
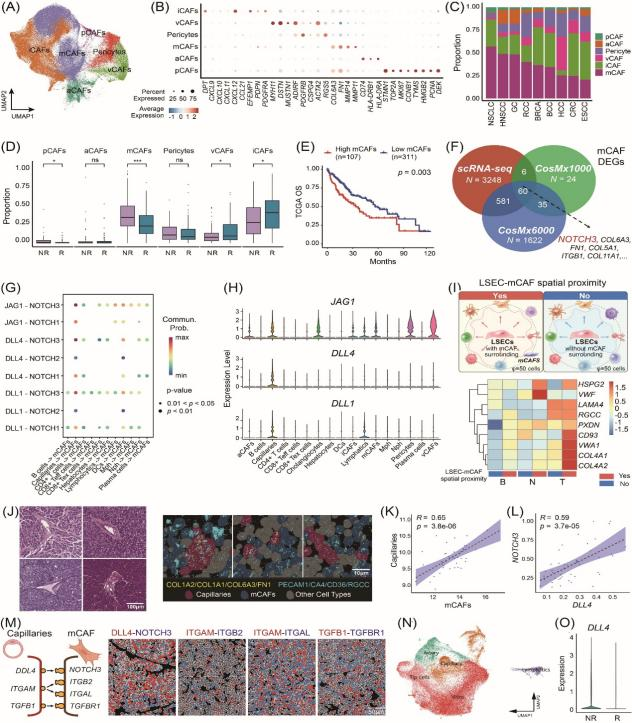
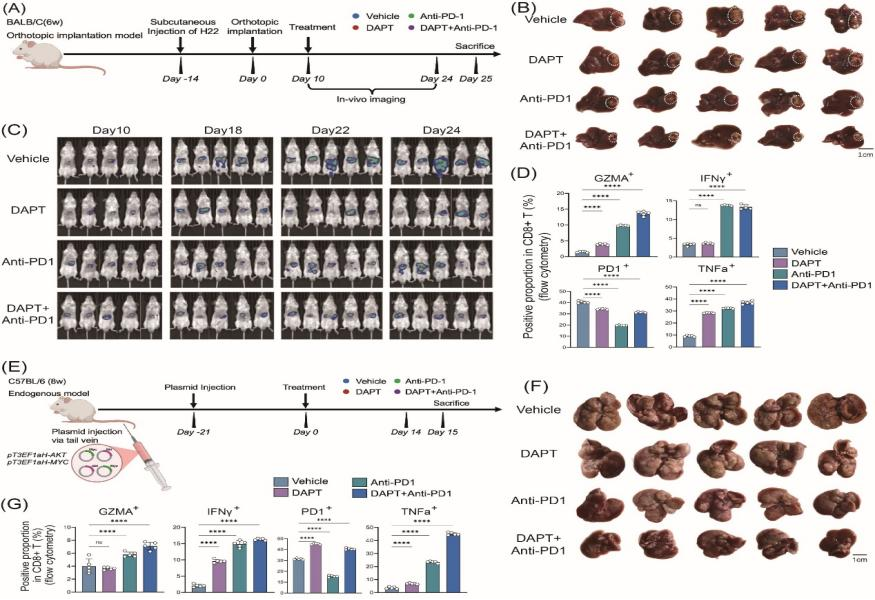
Mechanistically, NOTCH3 was identified as a key driver of collagen polarization and fibrotic deposition in mCAFs. Spatially constrained ligand–receptor interaction analysis revealed strong co-localization between DLL4 expressed by liver sinusoidal endothelial cells and NOTCH3 expressed in mCAFs, forming a specific vascular–fibroblast signaling unit. Activation of the DLL4–NOTCH3 axis promotes ECM remodeling and collagen crosslinking, increases tissue stiffness, and establishes both physical and signaling barriers that impede CD8⁺ T-cell infiltration into the tumor core. In vitro experiments showed that inhibition of NOTCH3 significantly reduced fibrotic phenotypes and enhanced CD8⁺ T-cell migration and cytotoxic activity. In both orthotopic and spontaneous HCC mouse models, pharmacological inhibition of the NOTCH pathway effectively disrupted the fibrotic barrier, increased intratumoral T-cell infiltration, and synergized with anti-PD-1 therapy to markedly reduce tumor burden.
This study provides important innovations at both mechanistic and translational levels:
Advancing spatial resolution of the HCC microenvironment: It offers a high-resolution characterization of fibrosis deposition and immune evasion landscapes, overcoming previous limitations in resolving key regions such as the invasive margin, and providing a reference framework for spatial cellular interactions.
Revealing a novel mechanism of immune exclusion: It identifies the DLL4–NOTCH3 axis mediating capillary–mCAF interactions, linking vascular signaling–driven fibrosis to immune exclusion and offering new insights into immunotherapy resistance.
Providing a translatable therapeutic strategy: It demonstrates the synergistic effect of NOTCH3 inhibition combined with anti-PD-1 therapy, supporting a novel “anti-fibrosis plus immunotherapy” treatment paradigm.
The study, titled “Spatial multi-omics identifies a NOTCH3-mediated capillary–mCAF crosstalk driving immune exclusion in hepatocellular carcinoma,” was published in iMeta. The work was conducted through collaboration between the Hepatobiliary and Pancreatic Center and the Department of Pathology at Beijing Tsinghua Changgung Hospital. Academician Dong Jiahong served as the corresponding author. Dr. Jifansen Ji, Dr. Haochen Li, and Dr. Qi Wang are co-first authors. This work was supported by major grants from the National Natural Science Foundation of China and Tsinghua University’s Precision Medicine Initiative, with additional contributions from collaborating teams.
